Full
FullGuide »
THIS SECTION CONSISTS OF DETAILS OF THE MAIN PESTS AND DISEASES IN COMBINING PEAS FOLLOWED BY FUNGICIDE AND INSECTICIDE CHECKLISTS, AND A PHOTO SECTION WITH REFERENCE IMAGES.
Many factors can affect growth of the pea crop, and the notes below describe the main pests and diseases which reduce yield and quality.
A wide range of localised climatic effects can influence the development of foliar diseases. In certain circumstances fungicides have been shown to be useful in controlling diseases. However, responses to treatment can only be expected if weather conditions favour disease development. Routine or prophylactic treatments do not produce an economic return year after year.
Weevil may cause damage if large numbers appear when plants are small and in particular in cloddy seedbeds and in conditions of slow growth.
Leaves of attacked plants show characteristic ‘U’ shaped notches around the edges, but the main damage occurs as a result of the larvae feeding on the root nodules. Sprays may be applied at the first sign of leaf damage and repeated after 7 - 10 days.
The adult weevils are beetle-like in shape and 4-5 mm in length. They are light grey to brown in colour with faint striping along the length of the wing cases. They have a very short ‘snout’ but have the ‘elbowed’ antennae typical of weevils.
Adults migrate from their over-wintering sites, mainly around the grassy uncultivated edges of fields previously cropped with peas or beans. Migration occurs early in the spring and this often coincides with short periods where the maximum air temperature exceeds 12°C.
During feeding, eggs laid by the female weevil are washed into the soil around the stem bases and produce larvae which begin feeding inside the root nodules. After 3-4 weeks, the larvae pupate and newly emerged adults return to the overwintering sites. There is one generation each year.
If leaf damage is anticipated, early treatment of the crop is necessary to interrupt the egg laying period and reduce root damage. A monitoring system is available which detects adults when they begin migrating in the early spring. The system was developed by Rothamsted Research and the field development was undertaken by PGRO in conjunction with Rothamsted Research and ADAS.
The monitoring system comprises five cone traps containing a pheromone lure. It can be used as an aid to decision making in the following ways:
Traps are sited on a single grassy verge or headland of a field which had been cropped with peas or beans the previous year. They should be sited by mid-February and weevils counted three times each week. Full instructions are supplied with the traps.
A threshold catch occurs when an average count per trap exceeds 30 weevils on any one recording day. Monitoring should continue until a threshold catch is reduced or until the latest sown crops have emerged (whichever is the sooner).
When a threshold has been reached, crops which have just emerged or will emerge during the next 10 days may be at risk. A spray can be applied as soon as the first sign of notching is observed if previous experience is that weevil damage occurs regularly. During periods of slow growth a second spray should be applied 10-14 days later. Crops emerging later should not be at risk. If a threshold is not reached or if it occurs more than 10 days before crop emergence there is no need to spray.
A monitoring system is available from Koppert UK to predict the likely severity of attack.
In most seasons, damage caused by thrips to newly emerging pea crops occurs to a greater or lesser degree. Attacks are more severe during periods of slow growth and in particular on stony soils.
Thrips are tiny, narrow-bodied, black insects of the type known as ‘thunderflies’. Many generations of thrips are wingless and spend most of the year in the soil, feeding on a wide range of non-legume crops including Brassicas, linseed and sugar beet. As peas begin to emerge in the spring, thrips feed inside the tightly rolled leaves of the growing point. Because feeding causes damage to the leaf surface, young leaflets appear pale and slightly distorted and, if held to the light, small translucent markings are obvious.
In many situations, peas can outgrow the initial attack, with no long-term effects on the crop. However, occasionally when the attack is severe, peas may develop multiple secondary shoots and develop as small bushy young plants. Where damage is noted in most years, then treatment can be justified.
Thrips in peas may be difficult to control as the insects feed inside the enclosed shoots. PGRO trials have shown that Hallmark with Zeon technology has given a useful and consistent reduction of leaf damage. The product is approved for use on all peas for weevil control. Early treatment is essential and where thrips damage is confirmed as the peas are just emerging, and before the leaves have expanded, a single spray should be applied as soon as possible.
The pathogen produces resting spores, which persist in the soil and initiate primary infection in young pea plants. Primary infected plants are stunted, covered on the underside of the leaves with grey coloured mycelial growth and often die before flowering. They are the source for secondary infection of older plants by producing air borne spores, especially under cool and humid conditions. Pods develop yellow blotchy patches on the surface. Seeds fail to develop properly and are often blemished.
Varietal resistance should be used to avoid serious losses. There are currently no fungicidal seed treatments for peas. There are no foliar fungicides which give effective control.
Attacks by this pest may result in loss of yield and can be very serious where populations have built up in intensive pea growing areas.
Description and life history
The adult, a small gnat-like insect, has a yellow-grey coloured body about 2mm in length, and six long legs. The head is dark with a pair of very fine antennae. The wings are semi-transparent and slightly longer than the body and when the midge is at rest, they are folded together along the insect's back.
Midges begin to emerge from previously infested fields during the first half of June, and after mating the females fly to those pea crops where buds and flowers are present. Batches of 20 or more eggs are laid on the young leaves surrounding the rudimentary buds, and on the buds themselves. After 4 days the eggs hatch and the larvae enter the developing buds where they live and feed at the base of the ovaries. The buds become swollen and gouty and do not produce pods, thereby resulting in loss of yield. Larvae may also feed in the clustered leaves of the growing point and the top of the plant remains shortened and develops a ‘cabbage’ or ‘nettle-head’ appearance. In wet periods, the damaged tissue may also provide a site for infection by fungi such as Botrytis spp.
After about 10 days, the larvae are mature and fall to the soil where some pupate and give rise to a second generation of adults 11 - 14 days later. Larvae produced by these adults, and those which do not pupate from the first generation, remain in the soil over winter. The majority emerge in the following two seasons.
Cultural control
There is little evidence of differences in susceptibility between varieties of peas although often it is the later sown crops which are affected. The prime sources of pea midge are those fields which carried heavily infested crops in the two previous seasons. Sowing peas in areas adjoining such infested fields should be avoided where possible. Crops in which the majority of flowering is over and where pods have set will not themselves suffer yield loss, but if attacked would contribute to the increase in the general level of midge population in the area. Large scale rotational practice involving neighbouring farms could be considered.
Chemical control
In areas of high incidence of attack, spraying of vining peas is recommended as soon as the first midges are seen in the crop. Vining pea crops from the Humber, northwards, may not suffer damaging attacks until late June and this means that in most areas, early crops need not be sprayed, but mid and late-season crops should be treated as they reach the susceptible stage.
Susceptible crops
At the early green bud stage, when crops will show their first white flowers in a weeks’ time.
Attacks by aphids may result in loss of yield by spoiling flowers, causing failure of pods to fill and by generally reducing plant efficiency. Aphids transmit viruses and produce honeydew, an ideal medium for colonisation by saprophytic fungi, in particular the sooty moulds. In addition, honeydew is very sticky and can lead to increased costs through the necessity of more frequent washing down of vining machinery.
Description and life history
The pea aphid (Acyrthosiphon pisum) is green in colour with a pear-shaped body, long legs and long antennae. There are both winged and wingless forms, the winged females being 2.5 – 3.0 mm long with deep red or black eyes, while the wingless females are of similar form but have a smooth, shiny outer skin.
Colonies are formed quickly, smothering the plant, as the female needs no male for fertilisation (parthenogenesis), and bears living young (viviparous) at a rate of 15 per day in suitable conditions. There are several generations in the pea aphid life-cycle. Eggs over-winter on forage crops such as lucerne and clover, hatching in the spring to give wingless stem mothers. These produce spring colonies of wingless females, which in turn produce winged females that move into the crop in summer. These migrants produce the colonies that damage the crop.
As the pea plants deteriorate the colonies produce winged female migrants that move to other plants to produce new colonies. Later in the season winged females move to over-wintering host plants where they produce males and females that lay eggs on the winter hosts.
Control
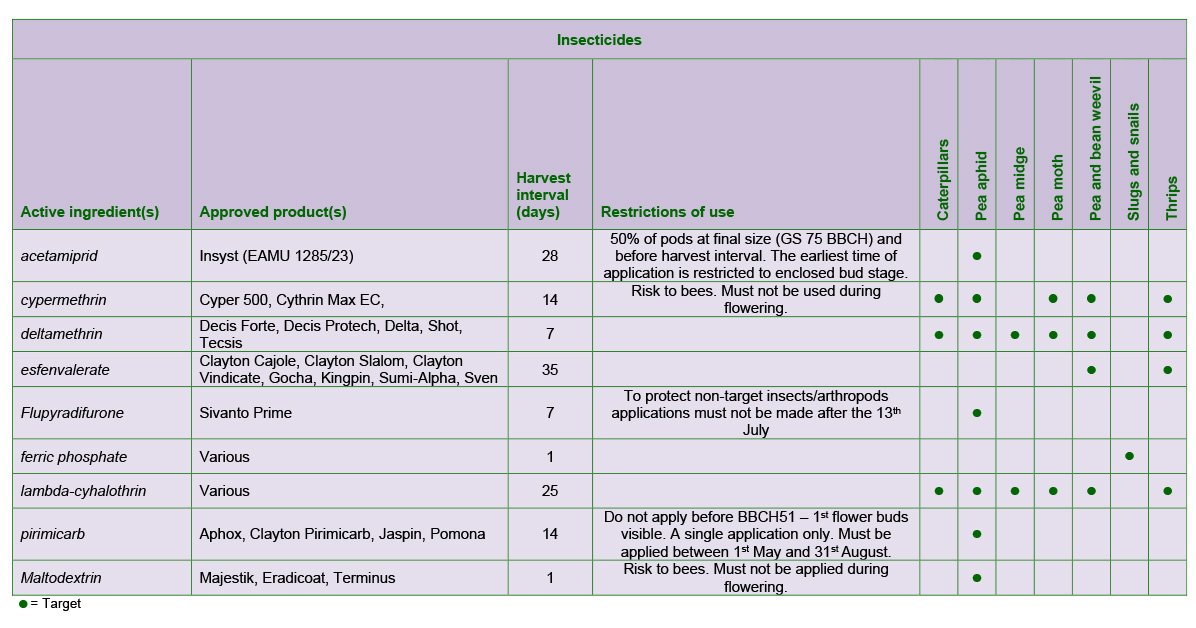
Losses due to aphids can be varied and considerable. If there is a light but general distribution in humid weather, or if breeding colonies are evident, then control measures are generally warranted. The thresholds for treatment of pea aphid to prevent direct feeding damage are as follows: spray combining peas when 20% of plants are infested and vining peas when 15% of plants are infested. Always check labels for harvest intervals.
The choice of product should be made with consideration of any other pests that are present at the time, such as pea midge and pea moth.
Timing of application can also affect the degree of virus infection as well as yield loss. In the case of pea seed-borne mosaic virus and pea enation mosaic virus aphids should be controlled as soon as they appear, even if the above thresholds are not met and particularly if this occurs before flowering. Where aphid infestation occurs late in the growing season yield increases from aphicides can be obtained in combining peas up to the development of the fourth pod-bearing node. Where infestation occurs after this point there is no appreciable yield loss.
The virus is primarily seed-borne but is transmitted by several aphids including the pea aphid, black bean aphid and peach-potato aphid. The virus becomes established after using infected seed and is transmitted by winged aphids early in the spring. Symptoms include vein-clearing, narrowing and downward rolling of leaflets and foreshortening of the apical internodes. Pods at the upper part of the plants may be stunted and appear to have a glossy green appearance. As the peas mature inside the pod a white blistering may develop giving the peas a ‘tennis-ball’ marking over the seed coat.
The virus affects quality in vining peas and it is important to maintain disease-free seed stocks. PSbMV can be detected in seed using an ELISA (enzyme linked immunosorbent assay) test, although not all infected seeds give rise to infected seedlings
The presence of this disease is seldom noticed before the approach of flowering, and often not until much later. PEMV is aphid-transmitted and is likely to have a greater effect on the crop if transmitted before flowering occurs. PEMV causes vein clearing and the formation of translucent spots which are apparent when infected leaves are held up to the light. Development of stipules is often retarded and they remain very narrow. Leaflets are crinkled and necrotic spots may appear. Often the tops of the plants become yellow and mottled with distorted leaves. Pods may be severely malformed and fail to fill.
The appearance of enations, small, irregular, protruding ridges of plant tissue which are found on the undersides of leaves and on pods, indicates an advanced stage of infection. Terminal growth ceases, axillary buds disappear and flower set is impaired. In severe cases yield is greatly reduced. Efficient control of aphids reduces the risk of losses.
Pea cyst nematode is a very persistent soil-borne pest, often causing severe yield loss. Frequent cropping of peas and Vicia faba beans favours the build-up of infestations, and an adequate rotation is essential to minimise the risk of occurrence. Affected plants are stunted and pale, and the root systems do not develop nitrogen-fixing nodules, but become studded with white, lemon-shaped cysts. Correct diagnosis is essential as subsequent pea crops grown in infested fields are subject to complete failure.
A deficiency or unavailability of the trace element manganese can cause a reduction of yield in combining peas, and, in addition, it may result in the appearance of marsh spot, which is liable to spoil the produce for human consumption and for seed purposes.
Occurrence and symptoms
The disorder is commonly associated with organic soils, although crops on other soil types can also be affected. Soil with a pH of more than 6.8 is likely to give a problem, especially in wet or compacted conditions, when manganese becomes unavailable to the plants and is "locked-up" in the alkaline soil. Over-liming must be avoided. Wheeling’s cause compaction and symptoms of manganese deficiency can often be seen as lines running through a crop.
Manganese deficiency causes a yellowing around the leaf edges and between the veins; entire leaves may become yellow in time, but the veins and the areas of tissue adjoining them are the last to lose their green colour, giving a somewhat striped appearance to the leaf.
Affected plants are likely to produce peas which have a brown and often granular area in the flat faces of the cotyledons, a condition which is known as "marsh spot". Such produce is unsuitable for human consumption, and for use as seed as resulting seedlings are often weak and malformed, with multiple shoots and short-lived growing points.
Treatments
Treat crops which are growing on land known to have a history of manganese deficiency and/or on land of pH 6.8 or more as routine, spraying at the 1st pod stage and again 10-14 days later. Crops in which symptoms are seen should be sprayed immediately and treatment repeated at the 1st pod stage. During wet weather flowering may be prolonged and a third application may be beneficial.
Treat with manganese sulphate (32% manganese) at 5 kg/ha in a high volume of water with an added wetter. Trials have shown that other products containing a high percentage of manganese are also effective. However, sprays with chelated forms (6% manganese) do not contain enough manganese to give adequate control.
Combined treatments
Sometimes, treatment against manganese deficiency coincides with treatment against pea moth or pea aphid or with a fungicide. Certain insecticides and fungicides can be mixed, but before doing so, the manufacturers of the products must be consulted.
Sclerotinia is favoured by warm, wet conditions. Infection occurs during flowering when sections of stems of individual or groups of plants become bleached and wilt or collapse. The fungus produces a fluffy white mycelium over the infection site. Pods can also become infected. Black, elongated sclerotia develop within the stem or on pods and these can contaminate the harvested produce.
Pea moth is one of the most damaging pea pests in this country and in Europe. The caterpillars (larvae) feed on peas within the pod. In vining peas, there is a risk of crop rejection because of contamination of the produce by damaged peas which cannot be removed mechanically. In combining peas for premium markets including human consumption or seed, damaged peas are removed by the merchant and the price paid to the grower is reduced in proportion. Although moth damage can reduce quality, the yield loss is rarely significant and the presence of damage in peas for animal feed compounding is not important. Control in these crops may only be justifiable where the damage levels in previous crops have been high.
Control
Spraying should be related to the development of the insect rather than to the stage of growth of the crop and therefore insecticides should be applied while the larvae are exposed, that is, from the time of hatching to the time of entering the pods. The timing of application is critical for maximum control.
Timing of sprays
Since pea moth can be a localised problem, overall spraying of peas over a wide area on any one date is not advisable as local conditions influence the behaviour of the pest. A system of accurately timing the application of insecticides is commercially available in the form of pheromone traps, combined with a simple model provided by PGRO to predict egg hatching. The traps, when placed in a pea field, attract male moths and by monitoring the catches in the trap, a grower can decide whether there is a need to spray, and time any necessary spray application efficiently on his own farm. The traps were developed by Rothamsted Research and fully tested by PGRO and ADAS.
Pea moth monitoring systems are available from the following suppliers:
Dragonfli, Unit 4 Rippers Court, Sible Hedingham, Halstead, Essex, CO9 3PY, UK Tel: 01376 563322 www.dragonfli.co.uk
Andermatt UK, 47 Compton Road, Brighton, West Sussex, BN1 5AL, UK Tel: 07939 395059 https://www.andermattuk.com
Koppert UK https://www.koppert.co.uk/products-solutions/
Traps are received in sets of one or two, depending on the supplier. One set is required for each block of peas, i.e. a farm having all the pea fields within a restricted area need only purchase one set of traps, but in fields of 50 ha or more two sets are required.
Traps must be placed in the pea crop by early May and examined at two-day intervals. The number of moths caught by each of them is noted on each occasion. Traps can be suspended on pheromone trap pole kits or fence posts in the field and should be placed at canopy height, moved upwards at intervals as the crop grows.
The threshold, which determines whether treatment is necessary, is ten or more moths caught in the single trap, or either of the two traps, on two consecutive occasions. Timing of sprays must be related to egg development and this is affected by temperature.
A spray date can be obtained from the pea moth page of the PGRO website - https://www.pgro.org/pea-moth/ based on a computer prediction, 3 - 4 days after reaching a threshold. On the predicted spray date, crops which are at the first pod set stage, or which have flowered should be sprayed, but later crops should only be sprayed when they reach first pod set. Crops with flat pods are susceptible to damage. A second application should be applied 10-14 days later
Severe infection usually occurs after a prolonged period of wet weather and may appear on crops at a slightly later stage than Botrytis. Often the disease is observed as a purple-brown flecking of the leaves and a more general browning or blackening of the stem, before lesions develop on the pod. The fungi are seed-borne and if infected seed is sown the following year, seedling losses, and later development of leaf and pod spotting, can result in more serious yield loss
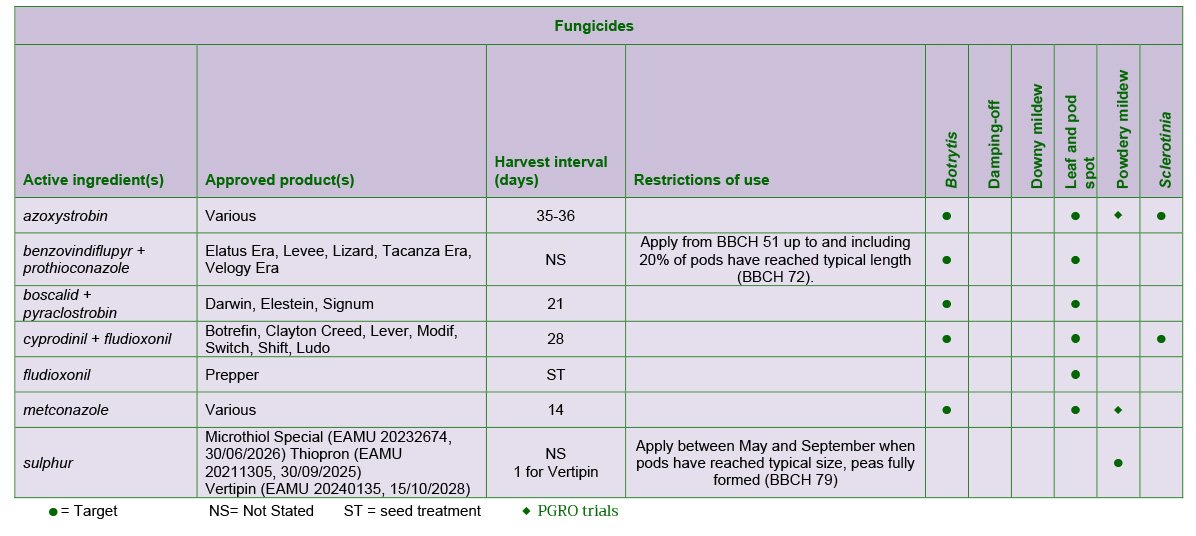
The most frequent is D. pinodes, which can cause losses in both yield and quality in wet conditions. The use of disease-free seed will help to reduce the incidence of disease. There are no minimum standards specified by the statutory seed certification scheme for D. pinodes but seed, especially farm-saved, should be tested. Fungicides such as azoxystrobin, metconazole and boscalid + pyraclostrobin give useful control of the disease in the crop and can give yield increases when applied during flowering and pod set.
The fungus is attracted first to wet flower petals which, after pod set, either become detached and lodge in the leaf axils or remain stuck to the developing pods. Once Botrytis comes into contact with green plant tissue via the rotting flower petal, it can penetrate and cause a rot of the stems or pods. Infected stems die prematurely, and pods may either abort, or the disease can rot the seeds within the pods and the pod itself. As well as a direct effect on yield, produce may be blemished, thereby reducing the quality of peas for processing or for seed.
One or two applications of fungicides at pod set and at the flat pod stage may be required to prevent Botrytis infection when wet or damp weather occurs during flowering. However, in dry conditions, sprays during flowering are unnecessary. Suitable products include boscalid + pyraclostrobin, cyprodinil
+ fludioxonil and azoxystrobin.
Occasionally late maturing crops may become covered with a grey-white film of powdery mildew. The disease can delay maturity but several commercial varieties are resistant to powdery mildew. The sulphur formulation, Thiopron (EAMU 0281/21), can be used to control powdery mildew.
A soil-borne disease which can occur in any pea growing area, but is generally confined to fields with a very long history of peas. It can cause substantial reductions in yield, but is effectively controlled by genetic resistance. Race 1 appears to be the most common form. The majority of varieties are resistant to this race and growers using land in known high risk areas should select these.
Information on pea foot and root rots can be found in the crop husbandry section of this guide. https://www.pgro.org/crop-husbandry/
Sclerotinia sclerotiorum causes a stem rot rather than a foot rot, but affects peas, spring beans, oilseed rape, linseed, and sometimes potatoes and certain field vegetables. This should be remembered when planning rotations in areas where Sclerotinia has occurred. Cyprodinil + fludioxonil or azoxystrobin applied at first pod can give useful control.
This is a potentially serious seed-borne disease, which can occur on all types of peas. Symptoms consist of water-soaked brown lesions on the lower leaves, stems and stipules, and become noticeable following periods of heavy rain, hail or frost. The lesions may coalesce and show a fan shape on the leaf, following between the lines of the veins. Some pod spotting may occur. Severe infections have not occurred in spring-sown peas and effect on yield has been negligible.
1. Every effort is made to ensure this information is correct. However, materials may be withdrawn, or there may be additional recommendations.
2. PLEASE READ & FOLLOW PRODUCT LABEL RECOMMENDATIONS CAREFULLY
Products containing potassium phosphonates (phosphites) are used to manage crop performance in both combining peas and field beans. However, residues of phosphonic acid are regularly detected in crops above the maximum residue levels. A recent publication in the EFSA journal desribes changes to maximum reside levels in several crops (EFSA Journal, September 2020, https://doi.org/10.2903/j.efsa.2020.6240), but legumes are not mentioned and there remains a risk that crops will be rejected for use where phosphonates have been applied, if they exceed maximum reside levels.